|
Circle the conjugate pairs and indicate the acid and base. Consider the species paired shown on the right. 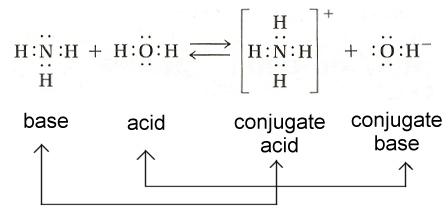
Write a balanced equation and indentify the conjugate pairs.ģ. Sulfuric acid, being a strong acid, the reaction proceeds in the forward direction until all the acid is used up. Sulfuric acid reacts in an acid base reaction with water with the transfer of one proton. Salts formed from those conjugate bases often have a pH of 7, or just above 7. The conjugate base of a strong acid is a very weak base. For example, the conjugate base of HCl is the chloride ion, Cl. Using the example above, that is ammonium, NH 4 . The conjugate \, acid, with the extra proton as it can donate that proton to another species. Using the example above, that is ammonia, NH 3. indicate with arrows if this reaction will proceed in the forward direction to a significant degree.Ģ. The conjugate base of an acid, is the ion that results when the acid loses a proton. The conjugate \, base, without the extra proton as the base is in a state to accept a proton. Electronegativity is one of those factors to think about. So when you're thinking about acid strength, think about the stabilization of the conjugate base and the different factors that can do that. Ammonia is a weak base and reacts with water according to the incomplete equation shown on the right. And if this is the most stable conjugate base, the fluoride anion is the most stable conjugate base, that means that H-F must be the strongest acid. In doing so, the acid becomes its conjugate base. The arrows indicate that the reaction will proceed to the right to a significant extent, as all strong acids and bases tend to do.ġ. In the Brnsted-Lowry system, an acid reacts by donating a proton to a base. The conjugate base can be determined by looking at the results of the chemical reaction. The acid base conjugate pairs for the reaction between nitric acid and hydroxide ion are: The base will always accept the proton while the acid will always donate the proton. A weak base will have a strong conjugate acid while a strong base will produce a weak conjugate acid. In the Brønsted–Lowry definition of acids and bases, a conjugate acid–base pair consists of two substances that differ only by one proton (H⁺).Ī conjugate acid is formed when a base accepts a proton while a conjugate base is formed when an acid donates a protonĮvery acid/base reaction consists of two conjugate acid/base pairs as shown on the right.Ī strong acid will have a relatively weaker conjugate base while a weak acid will have a relatively stronger conjugate base.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
